The Group is currently in the process of delivering the new inlays to at least one pharmaceutical company, but the RFID technology provider is also in discussions with other US-based businesses. The company began building RFID tag products for the pharmaceutical industry three years ago, according to Dean Frew, SML Group’s chief technology officer and senior VP of RFID solutions. However, he adds, widespread interest in the technology’s use for drugs and other controlled substances at hospitals didn’t see dramatic growth until 2019 and 2020.
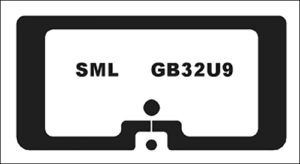
“Just like with every other vertical,” states Frew, “companies [in the pharmaceutical industry] are seeing what can be done with item-level RFID, and that leads to growing demand,” both for the tags and for solutions that utilize the tag-read data to improve operations. Another accelerator to that growth is the advent of a new standard for pharmaceutical RFID tags, created by the DoseID consortium (AIPIA newsletter 14/10/2020.) The GB32U9is among the first inlays to gain Spec S certification from the program.
The GB32U9, inlay is specifically designed for unit-of-use medications in pharmaceutical applications, leverages NXP’s latest chip, the UCODE 9. According to Frew, the inlay provides high quality and rapid inventory counting in dense RFID tag populations. The challenge, he says, was to design a tag that would not detune when attached to a liquid-filled product. The company created an antenna that responds reliably in the presence of liquids, as well as a variety of other materials, including plastics or glass. The antenna must also work despite its very small size.The inlay’s small design means it fits on a wide range of pharmaceutical products.
The GB32U9 was designed with auto-adjust technology, unique brand identifiers and a pre-serialized 96-bit Electronic Product Code (EPC). Its antenna measures 20mm by 10mm with a wet paper face size of 26mm by 12mm.Typically, the tags will be applied at the fill line by a pharmaceutical company, after which they can be read throughout the supply chain.
The small size and material challenges mean the read range is shorter than standard UHF RFID tags, with a range of about 1m, but that varies depending on the environment and the type of reader being used. However for most drug products this is not a major issue, says Frew. The inlay is available now, following approximately 18 months of engineering and testing.