Translumina, a developer and manufacturer of innovative cardiovascular medical devices used in interventional cardiology, announces the launch of Vivo Isar, its latest generation dual drug polymer-free coated stent (DDCS), in various international markets including Europe.
As an organization, Translumina has always been synonymous with innovation in the field of intervention cardiology and an announcement further strengthens its global cardiovascular portfolio.
Several studies have proven that polymers are pro-inflammatory and may cause stent thrombosis. However, the use of polymers has been necessary for current-generation DES as they control the release kinetics of the drug by acting as a carrier for drug loading and release.
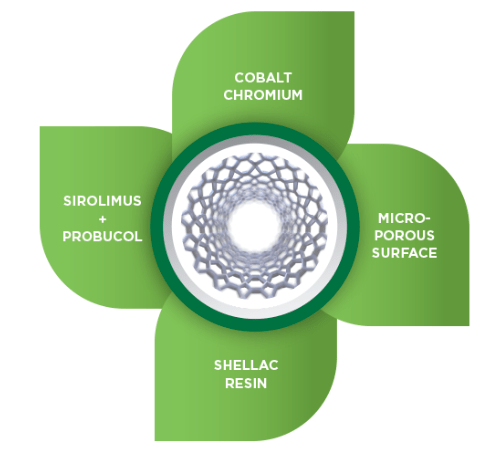
Vivo Isar is the first – dual drug polymer-free coronary stent technology, which brings together a stent with a superior safety profile without compromising the drug release kinetics. Vivo Isar is the first technology that uses probucol, an antioxidant and lipid-lowering drug, as a carrier for releasing Sirolimus (an anti-restenotic drug) and thus creates a polymer-free DES platform with uncompromised safety and efficacy.
In 2020, Translumina became the only company in the world to publish 10-year follow-up data on its dual-drug polymer-free coated stent (DDCS) Vivo Isar. The Journal of the American College of Cardiology (JACC), which is amongst the top publications in the world of cardiology, with a high impact factor as per Journal Citation Reports, published the 10-year safety and efficacy follow-up data on Vivo Isar.
“The ten-year outcome of Vivo Isar is a major breakthrough in DES technology as it proves that synthetic polymers can be removed from a DES without compromizing the efficacy of the DES. The excellent safety profile demonstrated in the 10-year follow-up study is especially notable as it provides powerful new technology for treating complex coronary artery disease in high-risk patient subsets. This DES technology carries the great potential of significantly shortening the duration of dual antiplatelet therapy,” said Dr Adnan Kastrati, director, Cardiac Cath Lab, German Heart Center, Munich and the lead investigator of Isar Test 5, the trial that compared Vivo Isar to Endeavour Resolute (Medtronic Inc USA).
Patients with diabetes mellitus present a particular challenge as the disease is associated with a more diffuse manifestation and consequently suboptimal clinical outcome after percutaneous coronary intervention.
In 2021, the Journal of American Heart Association published compelling 10-year efficacy and safety data of Vivo Isar in a Diabetes subset highlighting its excellent efficacy and safety profile, with a 30% reduction of MI as compared to Endeavour Resolute. Translumina became the first company in the world to achieve the longest RCT follow-up with its flagship dual drug-eluting stent which brings new hope to the high-risk diabetic subset.
Translumina has launched Vivo Isar in Italy and Spain and plans to roll it out in several markets across Europe, Latin America and APAC over the coming months.
“We are very pleased to be able to provide our premium high-quality stents to customers across the globe. Cardiologists world over now have a choice to give a proven and safer drug-eluting stent to their patients who otherwise have poorer outcomes with conventional DES, more so among the high-risk diabetic subset,” said Gurmit Singh Chugh, managing director and co-founder, Translumina.
Everstone Capital invested in Translumina in 2019 to further strengthen its research and development pipeline, scale its manufacturing operations and expand its global footprint to transform Translumina into a leading multinational medical devices company.
“We are proud that Translumina is leading the global journey of creating the highest standards of clinical data that enable physicians to treat patients with more confidence and certainty,” said Dr Arjun Oberoi, managing director, Everstone Capital.