Akiram Therapeutics, a Swedish biotech company specializing in radiotherapy presented promising preclinical data at the European Association of Nuclear Medicine (EANM) Congress taking place in Vienna, Austria on September 9-13. The data showcases the potential of the company’s radiopharmaceutical 177Lu-AKIR001 targeting tumor lesions for therapeutic purposes. A Phase 1 study is planned for 2024.
The 177Lu- AKIR001 is a new type of targeted radioimmunotherapy developed by Akiram Therapeutics. The therapy has shown promising results in several preclinical studies and holds the potential to become a first-in-class treatment. 177Lu-AKIR001 targets multiple cancer types, including anaplastic and iodine-refractory thyroid cancer, head and neck squamous cell carcinoma and non-small cell lung cancer.
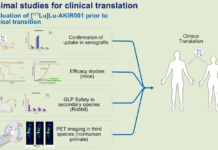
At the EANM Congress, Akiram presented findings from preclinical development work showcasing the utility of the radiotracer drug candidate, 177Lu-AKIR001, for therapeutic targeting of tumor lesions in a preclinical model of anaplastic thyroid cancer (ATC). The study aims to investigate the safety of 177Lu-AKIR001 in several species as well as the efficacy of the drug in preclinical models of ATC.
The results was presented by Dr. Anja Mortensen, co-founder and CTO on September 10 in Hall G2, session number 609.