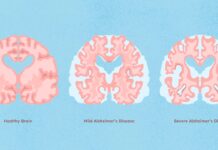
Eisai, a Japanese pharmaceutical company headquartered in Tokyo, and American multinational Biogen have released the results from their phase III clinical trial of lecanemab, showing that the drug can slow cognitive decline in patients suffering with Alzheimer’s disease.
Lecanemab is an investigational anti-amyloid beta protofibril antibody used to treat mild cognitive impairment that occurs due to Alzheimer’s disease and early Alzheimer’s disease with amyloid pathology in the brain.
The trial, conducted over 18 months, was a multi-center, double-blind trial including 1,795 patients from 50–90 years of age with early Alzheimer’s disease with amyloid, the CPHI online reported.
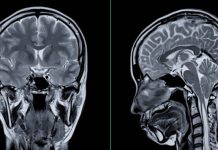
The efficacy of the drug was based on endpoints of a change in score on the Clinical Dementia Rating–Sum of Boxes (CDR-SB) from baseline, and a change in amyloid burden as shown by positron-emission tomography (PET).
In the case of both endpoints, lecanemab appeared to reduce the amyloid burden in early Alzheimer’s disease and slowed the decline in function and cognition according to the CDR-SB, compared to the placebo.
The results from the trial have been considered generally very positive. However, the researchers have stressed that the application of lecanemab was associated with adverse events, and in 14% of the treatment group, severe adverse events. These reactions included infusion-related reactions, ARIA with micro hemorrhages, atrial fibrillation, syncope, and angina. Deaths also occurred in 0.7% of the participants in the lecanemab group and 0.8% of the control group. The researchers highlight that the incidence of adverse events was similar between the test and control groups.
“All of these amyloid-lowering drugs carry a risk for an increased brain hemorrhage,” stated Ronald Petersen of the Mayo Clinic (MN, USA). “I think the primary outcomes, the secondary outcomes, the amyloid-lowering is pretty impressive.”
The data presented in the paper, published in The New England Journal of Medicine, also showed that according to the CDR-SB, those who had a genetic disposition to develop early Alzheimer’s disease did not show significant improvements with lecanemab.
The companies have applied for accelerated approval for the drug from the US FDA, hoping to be able to conduct further long-term studies and start treating patients, stating that the earlier the drug can be used in the disease stage, the more impact it will have on slowing cognitive decline.
“I believe it’s an important benefit that will justify full approval. But of course, we want a bigger benefit… before you’ve accumulated enough irreversible damage to be causing symptoms” commented Paul Aisen, Director of the University of Southern California Alzheimer’s Therapeutic Research Institute (CA, USA) and a co-author of the study.