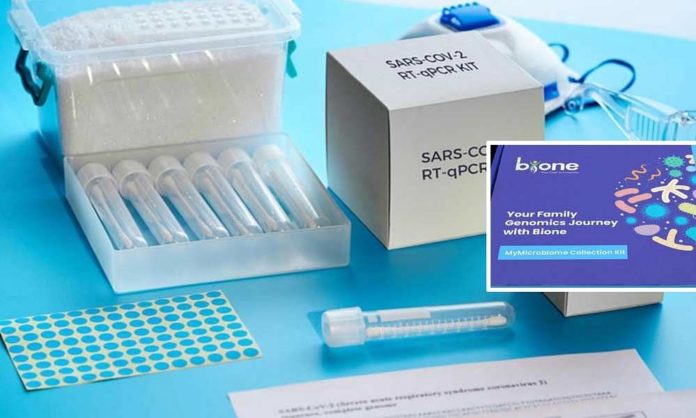
Following the news that India-based genetic and microbiome testing company Bione has received approval for its coronavirus (Covid-19) diagnostic test kit from the Indian Council of Medical Research (ICMR); Rohit Anand, medical devices analyst at GlobalData, offers his view, “As the Covid-19 cases in India are surging and the fear of community transmission is rising day by day, an urgent need for a large number of test kits to identify the suspected cases has arisen. However, India is continuing its approach to test patients with travel history or patients with severe symptoms due to the lack of testing kits.”
“Bione’s Covid-19 home screening test kits are expected to be available in the market shortly. Bione claims that its Covid-19 home rapid test kit can deliver accurate results within 5 to 10 minutes using blood sample with the help of alcohol swab and a lancet provided to prick finger. The company is said to be equipped to supply 20,000 test kits per week.
“GlobalData’s research reveals that the Indian in-vitro diagnostics (IVD) market, which accounted for more than 21% of the Asia-Pacific IVD market in 2019, is expected to grow at a compound annual growth rate of 2.5% through 2025.
“As the country will remain under complete lockdown for another two weeks, a home test screening kit is an effective way to identify the diseased population. The home rapid test kit is expected to release pressure on hospitals as they will be able to screen the diseased population quickly.”