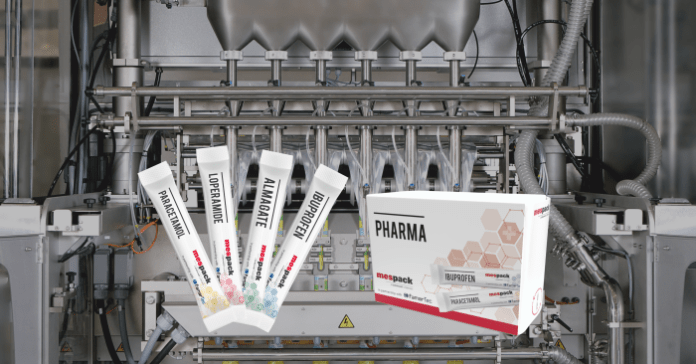
Mespack and Famartec have announced their collaboration regarding the development of a high-quality integrated solution for the pharmaceutical sector, which will integrate primary and secondary packaging. The growing demand from pharmaceutical customers for a complete solution to address their production needs encouraged both leading machinery manufacturing companies to join forces in order to respond to the market.
The joint project consists of a new vertical form-fill-seal machine for the production of stick-pack sachets, which are integrated into a carton module that groups the sachets before inserting them into the carton.
The line has been specially designed for the pharmaceutical industry and complies with specific regulations such as GMP, GMP5, and FDA’s 21 CFR Part 11. Regarding the pharmaceutics-focused documentation, this solution not only offers the execution of DQ, IQ, OQ, RA, but also the preparation of PQ and EN1020 standard 2.1 and 3.1 material certificates. Other types of documentation, upon the customer’s request, can be prepared as well.
Visitors will be able to view this special project at Mespack’s Interpack booth (B57-B73). The Interpack trade fair will be held from 4 to 10 May in Düsseldorf, Germany.
Mespack offers horizontal, including roll stock, pre-made, and hybrid), vertical, and water-soluble pods, equipment to produce sachets, stick packs, stand-up pouches or other film-based packaging, and end-of-line solutions.
Famartec has extensive knowledge of the design and manufacturing of automatic packaging machines for blisters, trays, cartons, and cases for pharmaceutical, nutraceutical, and cosmetic applications.